NIBRA-CS
USP: NIBRA-CS� breaks the barriers of traditional autonomic testing by making it accessible, affordable, and easy to use in everyday clinical practice. Unlike existing systems that are bulky, expensive, and dependent on laptops and specialized interpretation, NIBRA-CS� delivers instant, comprehensive reports at the point of care, significantly reducing the need for skilled man-hours and saving time for both clinicians and patients. Its portable, standalone design requires minimal infrastructure, making it deployable in small clinics, community health centers, and even remote settings where access to advanced diagnostics is scarce. By eliminating the dependency on heavy capital investment or advanced technical training, NIBRA-CS� brings autonomic testing within reach of a far wider healthcare network. This directly translates to cost savings for providers and more affordable testing options for patients. What truly sets NIBRA-CS� apart is its unique position as a nationally recognized, validated innovation. Backed by the Department of Science and Technology DST and the Department of Biotechnology DBT , licensed by CDSCO, and highlighted as a success story at Global Bio-India, it carries both credibility and clinical validation. With over 500 patients tested in research studies, its proven efficacy has already contributed to advancing CAN research in India. Launched on the global stage at Global Bio-India 2023, NIBRA-CS� has emerged as a pioneering diagnostic tool�an indispensable solution that redefines the standard of care in autonomic neurofunction testing.
- Innovator's Name
- Product/Technology Description
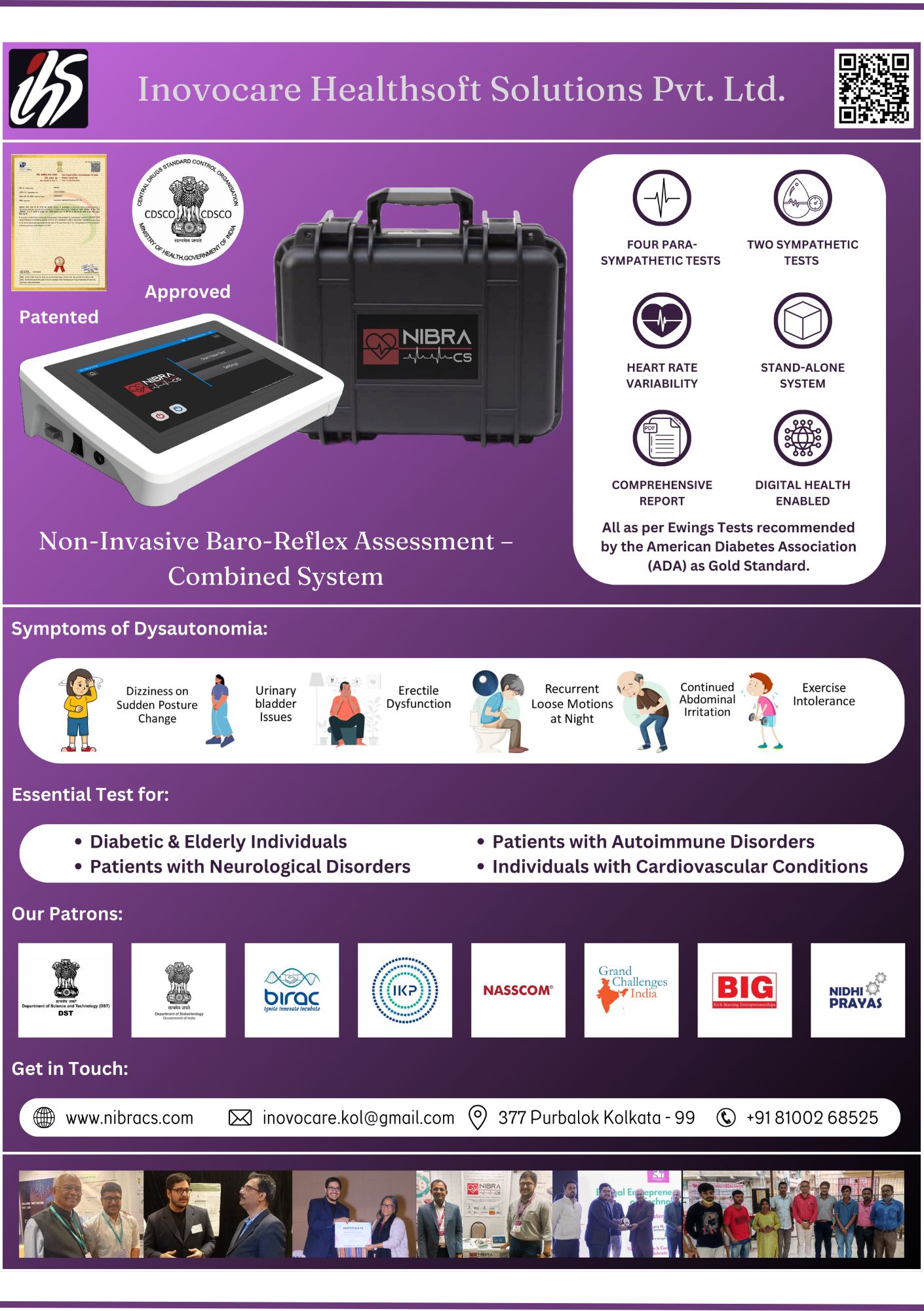
- Product Name: NIBRA-CS
- Product Title: Non-Invasive Baro Reflex Assessment - Combined System
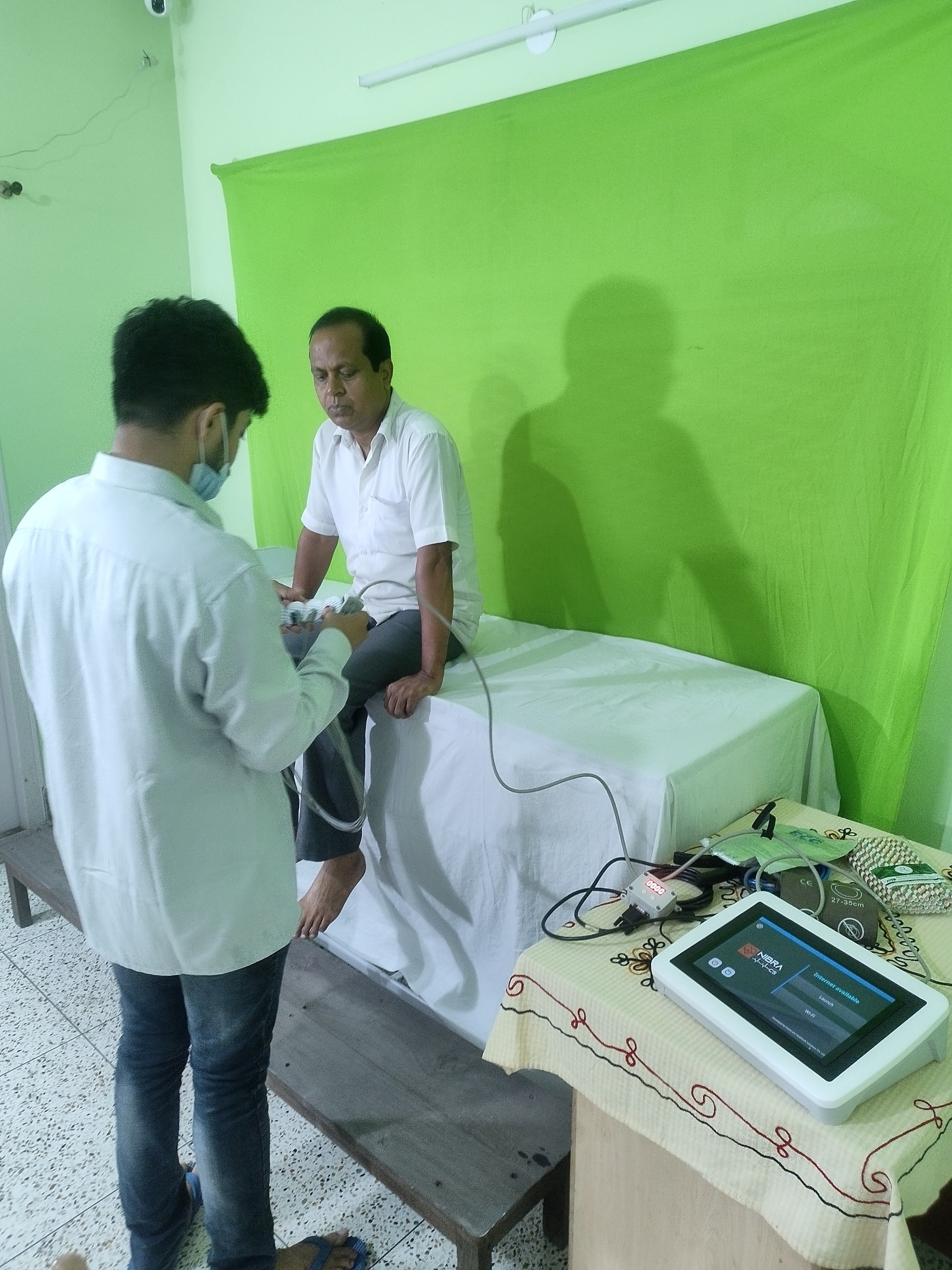
- Description: NIBRA-CS� Non-Invasive Baro-Reflex Assessment � Combined System is a patented diagnostic unit designed for autonomic neurofunction testing based on Ewing�s Battery of Tests, the globally recognized gold standard. The device evaluates autonomic responses to standardized physiological stimuli�such as deep breathing, posture changes, and Valsalva maneuver�through non-invasive monitoring of cardiovascular parameters. By analyzing heart rate variability HRV and baroreflex sensitivity, it provides a comprehensive picture of autonomic nervous system function. Unlike conventional setups, NIBRA-CS� is portable, digital-health enabled, and fully standalone, requiring minimal infrastructure. It is tailored for point-of-care settings, enabling early detection of autonomic neuropathy, a complication highly prevalent yet underdiagnosed among diabetic patients, the elderly, and even long-COVID survivors. The American Diabetes Association ADA recommends annual autonomic function screening from the time of type 2 diabetes diagnosis, underscoring the clinical urgency NIBRA-CS� addresses. Recognized with multiple national grants AGC JanCare, BIRAC BIG, NIDHI Prayas, TIDE 2.0 , NIBRA-CS� received its CDSCO Test License and was officially launched at Global Bio-India, Pragati Maidan, New Delhi, by BIRAC, Department of Biotechnology, Government of India. The solution is currently being piloted for PHC-level integration with NASSCOM Foundation. Website: www.nibracs.com
- Unique Selling Point: NIBRA-CS� breaks the barriers of traditional autonomic testing by making it accessible, affordable, and easy to use in everyday clinical practice. Unlike existing systems that are bulky, expensive, and dependent on laptops and specialized interpretation, NIBRA-CS� delivers instant, comprehensive reports at the point of care, significantly reducing the need for skilled man-hours and saving time for both clinicians and patients. Its portable, standalone design requires minimal infrastructure, making it deployable in small clinics, community health centers, and even remote settings where access to advanced diagnostics is scarce. By eliminating the dependency on heavy capital investment or advanced technical training, NIBRA-CS� brings autonomic testing within reach of a far wider healthcare network. This directly translates to cost savings for providers and more affordable testing options for patients. What truly sets NIBRA-CS� apart is its unique position as a nationally recognized, validated innovation. Backed by the Department of Science and Technology DST and the Department of Biotechnology DBT , licensed by CDSCO, and highlighted as a success story at Global Bio-India, it carries both credibility and clinical validation. With over 500 patients tested in research studies, its proven efficacy has already contributed to advancing CAN research in India. Launched on the global stage at Global Bio-India 2023, NIBRA-CS� has emerged as a pioneering diagnostic tool�an indispensable solution that redefines the standard of care in autonomic neurofunction testing.