The aim is to establish Network of Oncology Clinical Trials in India (NOCI), which will enhance the capability for conduct of Industry-sponsored trials of new cancer drugs and biosimilars by: Manpower training and infrastructure development, process harmonization between centres and to conduct collaborative investigator - initiated research. The purpose is to strengthen Clinical Trial capacity and to create a registry of common cancers which gives background data on outcomes. The network brings 6 large oncology centres representing different regions of India (North 2, South 3 & East 1) with access to large patient pool and increase their clinical trial capabilities. Jawaharlal Institute of Postgraduate Medical Education and Research (JIPMER), Puducherry is lead site of the network. An individual patient data registry of cancer care and outcomes being developed in collaboration with six other centres across India.
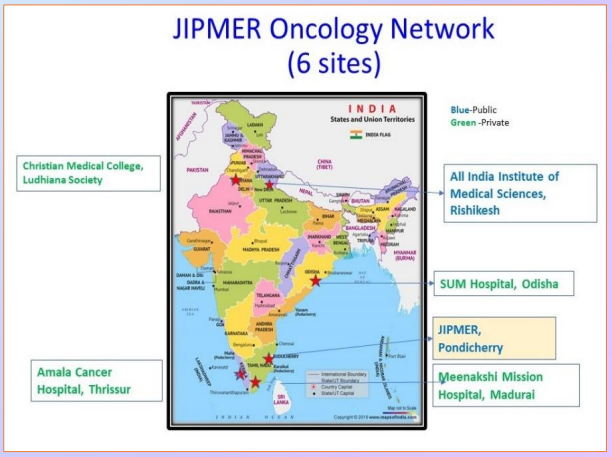
A common protocol for establishing the database has been established among all centres and approved by Institutional Ethics Committee. Site started creating database for 06 common cancers (Lung, Head and neck Cervix, Ovary, Breast and Brain cancers) where data of patients are captured to a software aiming to aid in real-world evidence of disease manifestation and treatments.